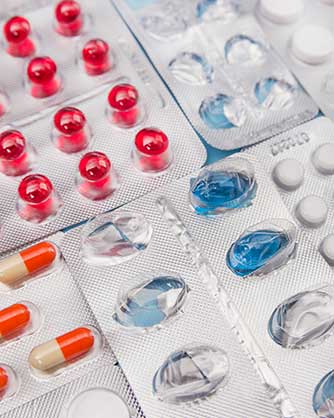
Innovating the Future of Pharma Manufacturing
For over decade, we’ve invested in cutting-edge facilities and technology, earning WHO cGMP certification which reflects the sophistication of our manufacturing units.
Our commitment to Total Quality Management (TQM) across all stages, along with Good Laboratory and Clinical Practices (GLP, GCP), ensures world-class products and services.
We offer a comprehensive range of manufacturing capabilities, with ample capacity for producing nasal sprays, ampoules, vials, tablets, capsules, liquids, ointments, and dry syrups. Our advanced machinery, reliable infrastructure (including purified water, CNG gas, and consistent power supply), and dedication to continuous improvement make us the ideal outsourcing partner pharmaceutical companies.
The company has adequate capacity in:
- Nasal Drops/ Spray : 20 million bottles
- Ampoules : : 40 million
- Vials : 20 million
- Tablet: 500 millions
- Capsules: 120 millions
- Liquid: 10 millions bottles
- Ointment: 6 millions tubes
- Dry syrup : 3 million bottles
Our track record speaks for itself, with satisfied clients across the globe in 70+ countries. State-of-the-art quality control and R&D departments uphold our commitment to international pharmaceutical standards.
At every step, from raw material sourcing to finished product, we prioritize quality. We constantly update our knowledge and processes to maintain our position as a leading provider of world-class pharmaceutical manufacturing & products.
Explore Our Core Manufacturing Categories
Explore Our Core Manufacturing Process

Formulation Development
Transforming ideas into safe, effective, and scalable pharmaceutical products.

Bulk Manufacturing

Regulatory Compliance & Quality Assurance
Why Choose prazky for Pharmaceutical Manufacturing?

High Compliance Standards
Innovative Technology Integration


